022-29398436
Agriculture, forestry, animal husbandry and fishery, industrial and mining enterprises, municipal engineering,hydrology
and water conservancy, scientific research institutions, real estate and construction industry, etc

The common valence states of chromium compounds are trivalent and hexavalent. In water, hexavalent chromium is generally in the form of cr2o72-, cro42- and can be transformed into each other by the influence of pH value, temperature, redox substance and organic matter.
Chromium is one of the necessary trace elements in organism. The toxicity of chromium is related to its valence state. Hexavalent chromium has strong toxicity, which is a carcinogen and is easily absorbed by human body and accumulated in vivo. It is generally believed that hexavalent chromium is 100 times more toxic than trivalent chromium. However, for fish, trivalent chromium compounds are more toxic than hexavalent chromium. When the concentration of hexavalent chromium in water reaches lmg/l, the water is yellow and astringent; When the concentration of trivalent chromium reaches lmg/l, the turbidity of water increases obviously. The natural water on land generally contains no chromium; The average concentration of chromium in seawater is 0.05 μ G / L, lower drinking water.
The industrial pollution sources of chromium mainly come from the waste water of chromium ore processing, metal surface treatment, leather tanning, printing and dyeing, photographic materials and other industries. Chromium is an important index of water pollution control.
The determination methods of chromium in water mainly include diphenylcarbazide spectrophotometry (gb/t 7467-87), atomic absorption spectrophotometer, ammonium ferrous sulfate titration, etc. Spectrophotometry is a standard method at home and abroad; Titration method is suitable for water samples with higher chromium content.
Determination of hexavalent chromium
In acidic medium, hexavalent chromium reacts with diphenylcarbazide (DPC) to form a purple red complex, which is determined by colorimetry at 540nm. The reaction formula is as follows
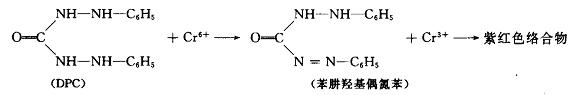
The minimum detectable concentration of this method is 0.004 mg / L, and the upper limit of determination is 1 mg / L with 10 mm cuvette. The main points of determination are as follows
(1) For clean water samples, it can be determined directly; For the water sample with low chroma, the blank water sample with acetone instead of chromogenic agent can be used for reference determination; For turbid and deep color water samples, zinc hydroxide is used as coprecipitating agent, and the pH of the solution is adjusted to 8-9. At this time, Cr3 +, Fe3 +, Cu2 + all form hydroxide precipitation, which can be removed by filtration and separated from Cr6 + in the water samples. When there are reducing substances such as sulfite and ferrous chloride and oxidizing substances such as hypochlorite, corresponding interference elimination measures should be taken.
(2) Take an appropriate amount of clean water sample or pretreated water sample, add acid, develop color, fix volume, use water as reference, measure its absorbance and make blank correction, check and calculate the content of hexavalent chromium in water sample from the standard curve.
(3) Prepare a series of chromium standard solutions and operate according to the water sample determination steps. After the measured absorbance was corrected by blank, the standard curve of absorbance to hexavalent chromium content was drawn.
Spectrophotometric Determination of diphenylcarbazide
Analyte: Hexavalent chromium
Measurement range: 0-1ppm (range extendable)
Not yet



